Clinical Study Protocol Template - Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. This template aims to facilitate the development of phase 2 and 3. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich. Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as.
Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). This template aims to facilitate the development of phase 2 and 3. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich. 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as.
Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). This template aims to facilitate the development of phase 2 and 3. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as. Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch.
TEMPLATE CLINICAL STUDY PROTOCOL
Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as. This template aims to facilitate the development of phase 2 and 3. Investigators for such trials are strongly encouraged.
Clinical Trial Protocol Template Word
5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich. This template aims to facilitate the development of phase 2 and 3. Investigators.
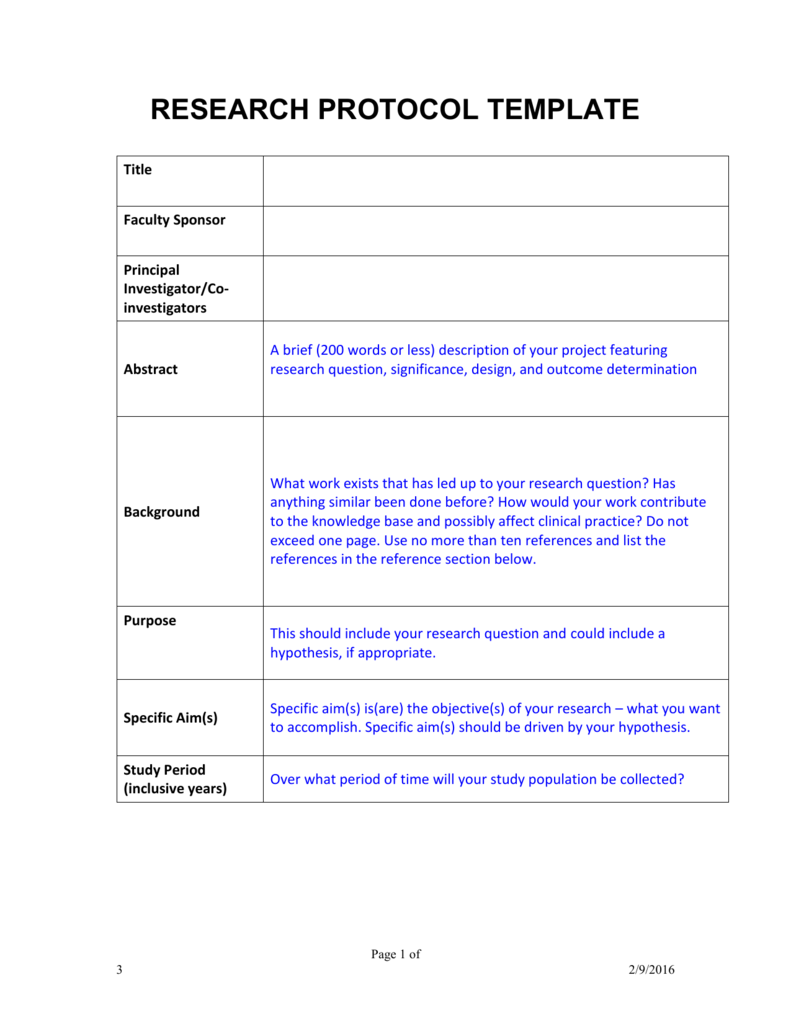
research protocol template
5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as. Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following.
Clinical Study Protocol Template prntbl.concejomunicipaldechinu.gov.co
This template aims to facilitate the development of phase 2 and 3. Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. Nih applicants can use a template with instructional and.
Clinical Study Protocol PowerPoint and Google Slides Template PPT Slides
Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be.
Clinical Study Protocol Template prntbl.concejomunicipaldechinu.gov.co
Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich. Investigators for such trials are strongly encouraged to use this template when.
Clinical Study Protocol PowerPoint And Google Slides, 53 OFF
This template aims to facilitate the development of phase 2 and 3. 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich. Nih.
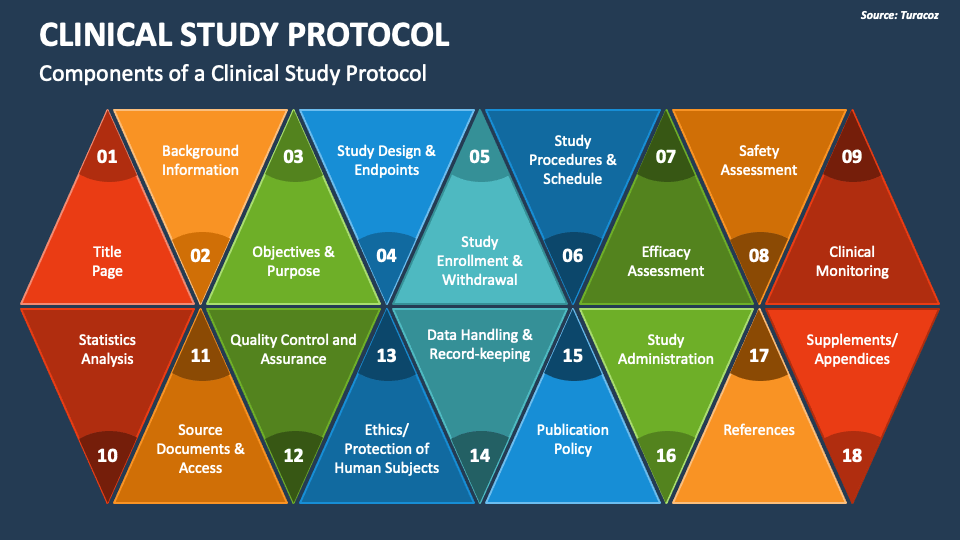
Clinical Study Protocol (CSP) Template Clinical Study Templates
5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as. Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). This template aims to facilitate the development of phase 2 and 3. At step 2 of the ich process, a.
Prescribing Protocol Template for New Drugs
Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as..
Clinical Study Protocol Template
This template aims to facilitate the development of phase 2 and 3. Phase iiib study clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich. Nih applicants.
At Step 2 Of The Ich Process, A Consensus Draft Text Or Guideline, Agreed By The Appropriate Ich Expert Working Group, Is Transmitted By The Ich.
Investigators for such trials are strongly encouraged to use this template when developing protocols for nih supported clinical trial(s). 5 this template is intended for interventional clinical trials of drugs, vaccines, and drug/device 6 combinations intended to be registered as. This template aims to facilitate the development of phase 2 and 3. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research:.